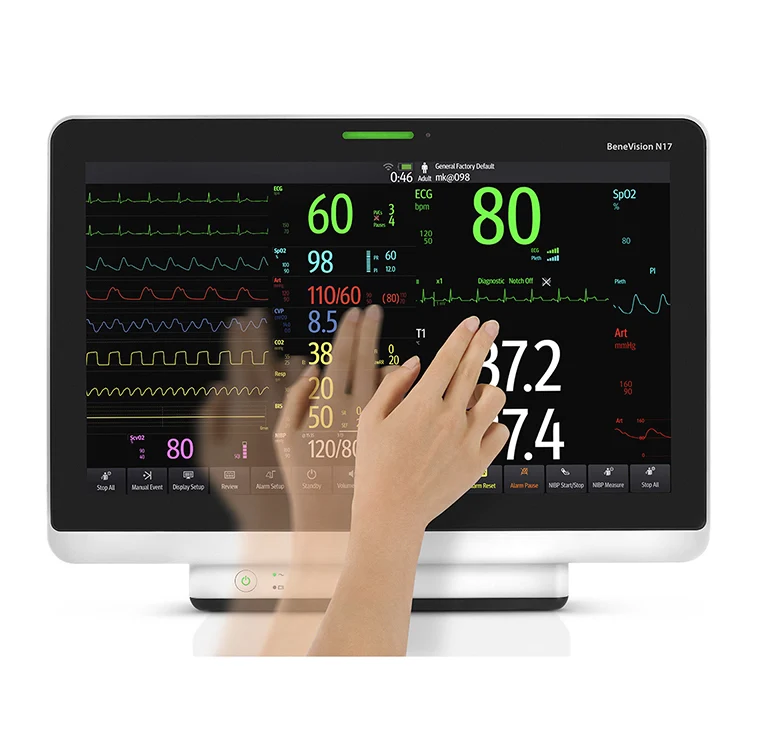
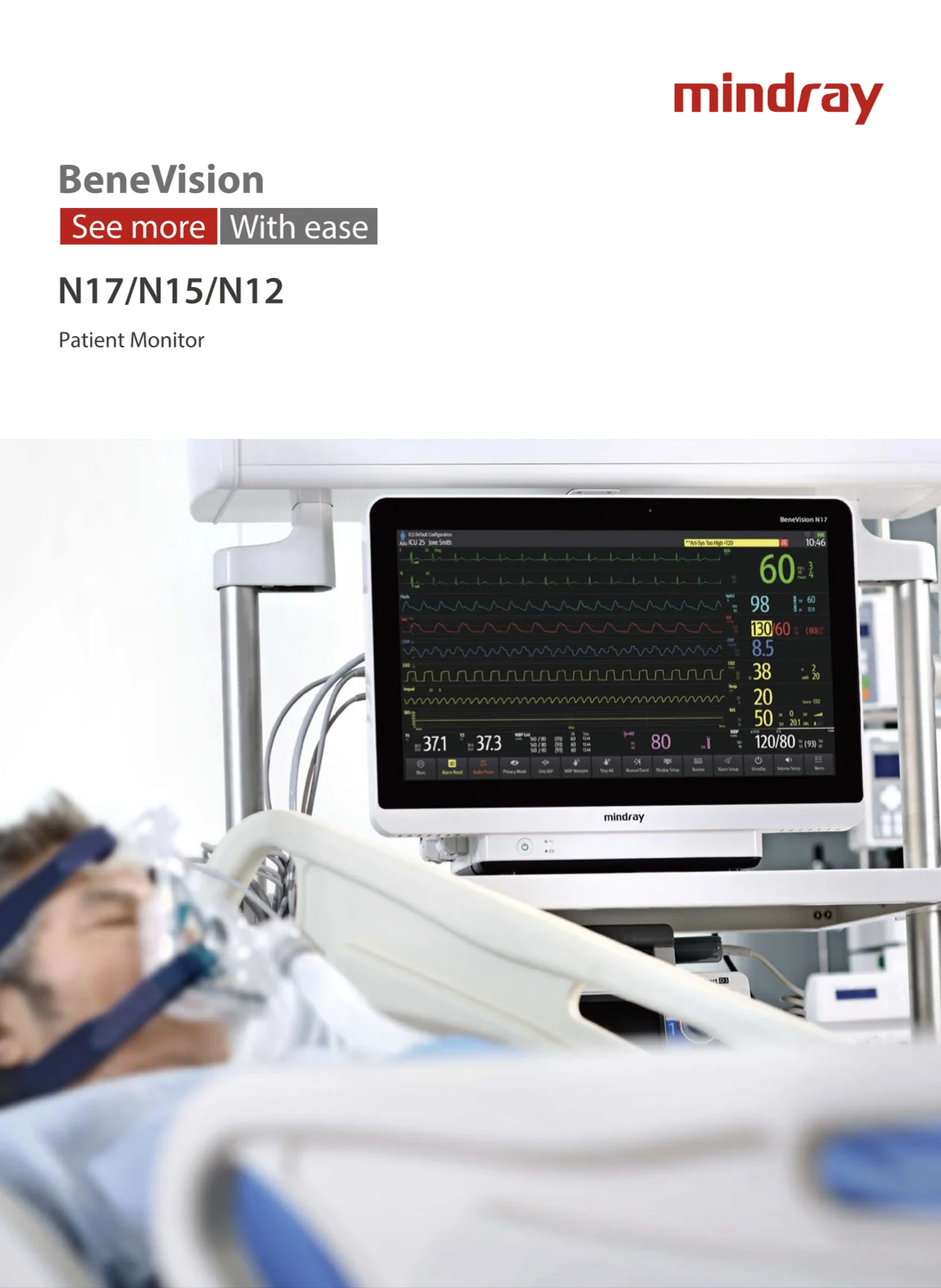
Designed to improve clinical safety and efficiency through operational design, the N Series range represents the future of patient monitoring, offering greater visibility and consistency of care throughout the patient journey:
N12: 12.1inch WXGA touchscreen display with up to 8 traces
N15: 15inch full HD touchscreen display with up to 10 traces
N17: 18.5inch full HD touchscreen display with up to 12 traces